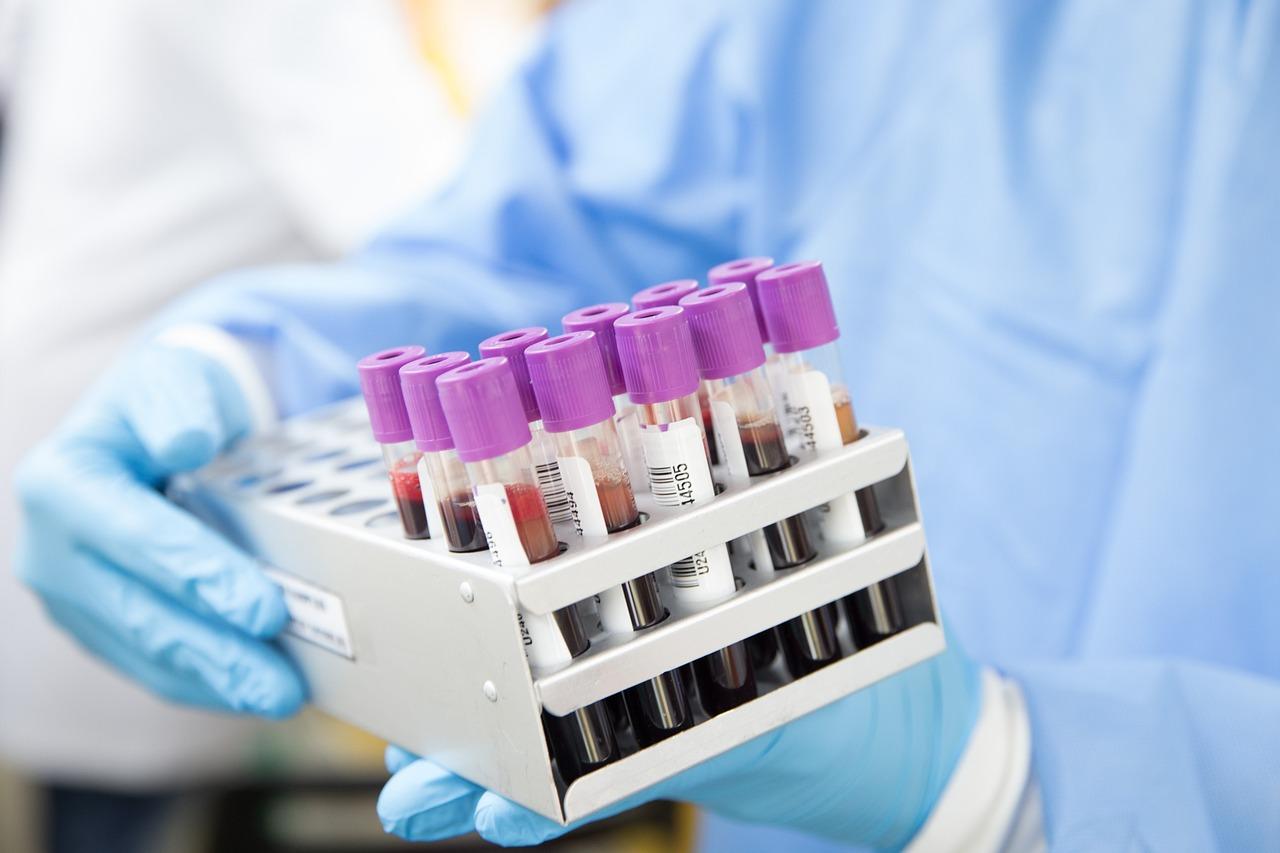
Blood typing-related products mainly include two sectors of blood typing instruments and blood typing reagents.

The instruments employed by hospitals participating in the basic study were mainly from various manufacturers such as Sysmex, Stago, Werfen, Sekisui, Beckman, Mindray, Succeeder, BSBE, and Rayto, involving 96 instruments, of which 11 hospitals used instruments from two manufacturers, with the use of each brand of blood coagulation analyzers shown in Fig. 4.

Truvian Health (“Truvian”), a diagnostics company redefining routine blood testing, today announced U.S. Food and Drug Administration (FDA) clearance for the Complete Blood Count (CBC) on its TruVerus™ multi‑modal blood testing system (K251249).

The biggest difference between “dry chemistry” and traditional “wet chemistry” (i.e., solution chemistry) is the media involved in the chemical reactions.

Leica Biosystems, a cancer diagnostics company and a global leader in workflow solutions, and Molecular Instruments® (MI), the inventor of the HCR™ Imaging technology, today announced a partnership to enable fully integrated RNA-ISH using the BOND RX and BOND RXm research staining systems.

Database building is a link to high-throughput sequencing.

The miniaturization of fluorescent PCR instruments is also a trend. Small PCR cyclers are portable and realize mobile detection.

In 2020, NMPA approved 18 types of class III molecular diagnostic instruments, and the provincial medical product administration approved 11 types of class II molecular diagnostic instruments, a total of 29.

QIAGEN (NYSE: QGEN; Frankfurt Prime Standard: QIA) today announced it has entered into a collaboration with Eli Lilly and Company to support the development of a QIAstat-Dx in-vitro diagnostic (IVD) to detect APOE genotypes which can play a role in the diagnosis of Alzheimer’s disease. This collaboration represents a significant milestone as the QIAstat-Dx panel would be the first commercially available IVD for APOE genotyping.

Building a complete biochemical testing system is the future development trend in the biochemical industry.
✔ All (26)
✔ Press release (0)
✔ Industry news (26)
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
