


Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Themed "Global IVD Innovation & Cross-Regional Integration," this groundbreaking event brings together distinguished experts from around the world to explore the future of diagnostics.
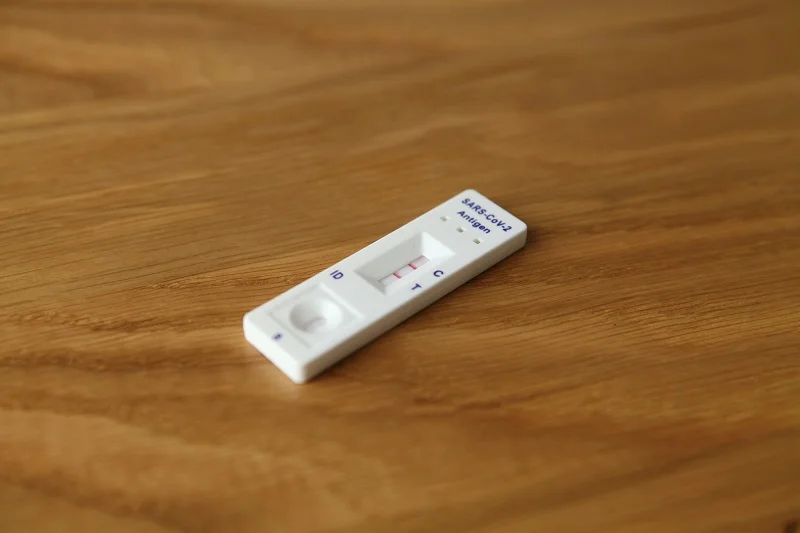
Jiangsu Huadong Medical Device Industrial and Getein Biotech have signed an agreement to jointly manufacture and sell antigen test kits for Covid-19 and other in vitro diagnostics (IVD) products.

On January 9, Fosun Foundation, together with Fosun Pharma and Henan Genuine Biotech Co., Ltd., announced that they will donate 100 million yuan worth of oral drug Azvudine to rural areas in China.

Becton, Dickinson and Company (BD) and CerTest Biotec have announced that their molecular assay for detecting the Mpox virus has received Emergency Use Authorization (EUA) from the US Food and Drug Administration (FDA).

QIAGEN today announced it has completed the acquisition of Verogen, a leader in the use of next-generation sequencing (NGS) technologies to drive the future of human identification (HID) and forensic investigation.

On January 5, 2023, Guangzhou Darui Biotechnology Co., Ltd., the subsidiary of Daan Gene Co., Ltd., received China National Medical Products Administration (NMPA) class III medical device approval for its “Twenty Genetic Deafness Genes Mutation Detection Kit (Time of Flight Mass Spectrometry)”.

Genomic and diagnostic testing company Prenetics Global said Tuesday that the US Food and Drug Administration has cleared ACTOnco, a comprehensive genomic profiling test for solid tumors.

Foundation Medicine and Karyopharm Therapeutics said Tuesday they are collaborating on a companion diagnostic for Karyopharm's experimental drug Xpovio (selinexor), which is in development as a maintenance therapy for patients with advanced or recurrent TP53 wild-type endometrial cancer.

January 10, Suzhou Nanomicro Technology Co., Ltd. issued a voluntary disclosure announcement for its 2022 annual results forecast.

Foundation Medicine, Inc., a pioneer in molecular profiling for cancer, and Natera, Inc., a global leader in cell-free DNA testing, today launched an early access program for clinical use of FoundationOne®Tracker, a personalized circulating tumor DNA (ctDNA) monitoring assay.

Being committed to continued advances in improving human health, SCHOTT has opened a new facility in Phoenix, Arizona, to expand the development and manufacturing of diagnostics and life science consumables.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
