


To ensure the quality and safety of medical services in primary healthcare institutions, China’s National Health Commission (NHC) and other relevant authorities issued a notice on 1 April 2026, outlining a three-year nationwide action.

Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.
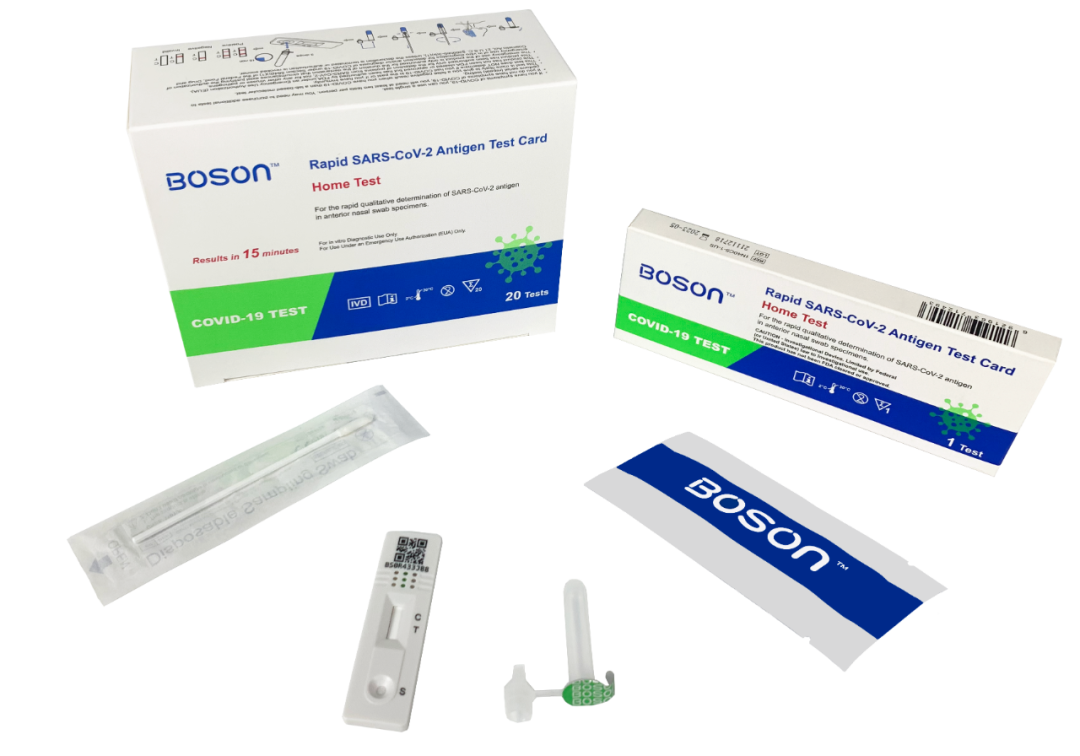
On April 6, it was reported that the Rapid SARS-CoV-2 Antigen Test Card produced by Xiamen Boson Biotech Co., Ltd. has obtained the FDA Emergency Use Authorization (EUA).

Seegene said on Wednesday that it has received Australian approval and CE-IVD marking for its Allplex RV Master Assay for respiratory viruses.

April 5, 2022 – GE Healthcare and Elekta (EKTA-B.ST) announced today that they have signed a global commercial collaboration agreement in the field of radiation oncology.

On 28 March, Sanxing Medical Electric Co., Ltd. (601566.SH), made several announcements announcing that its subsidiary, Rehabilitation Investment, intended to acquire 100% of the equity interests in five hospitals for a total price of CNY 844 million.

UK life science tools company LGC said on Friday that it has acquired Rapid Genomics, a next-generation sequencing genotyping company based in Gainesville, Florida.

Agilent Technologies on Tuesday announced it has obtained expanded CE-IVD marking for the use of its PD-L1 IHC 28-8 pharmDx immunohistochemical assay as a companion diagnostic test to guide treatment for newly approved indications for Bristol Myers Squibb's Opdivo.

Jinan Babio Biotechnology Co., Ltd. recently received an official notification from the EU Notified Body for its self-tested novel coronavirus antigen detection product, and subsequently obtained the CE certification issued by PCBC.

Thermo Fisher Scientific’s new Gibco CTS Xenon Electroporation System aims to provide easier scale up for cell therapies, from clinical development to commercial manufacturing.

Israeli diagnostics firm Todos Medical on Friday reported a nearly 13 percent year-over-year increase in revenues for the fourth quarter on increased demand for its COVID-19 testing products and services, as well as nutritional supplements.

On March 30, China National Medical Device Co., Ltd. (CNMD for short) under Sinopharm Group and Suzhou Hybiome Biomedical Engineering Co. Ltd., subsidiary of BioMerieux in China held a signing ceremony of the strategic cooperation agreement.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
