


To ensure the quality and safety of medical services in primary healthcare institutions, China’s National Health Commission (NHC) and other relevant authorities issued a notice on 1 April 2026, outlining a three-year nationwide action.

Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Hurun Report released the Hurun Global Rich List 2022, a ranking of the US dollar billionaires in the world. Wealth calculations are a snapshot of 14 January 2022. This is the eleventh year of the ranking.

Burning Rock Biotech reported late Monday that its fourth quarter 2021 revenues were up approximately 12 percent compared to the same quarter last year
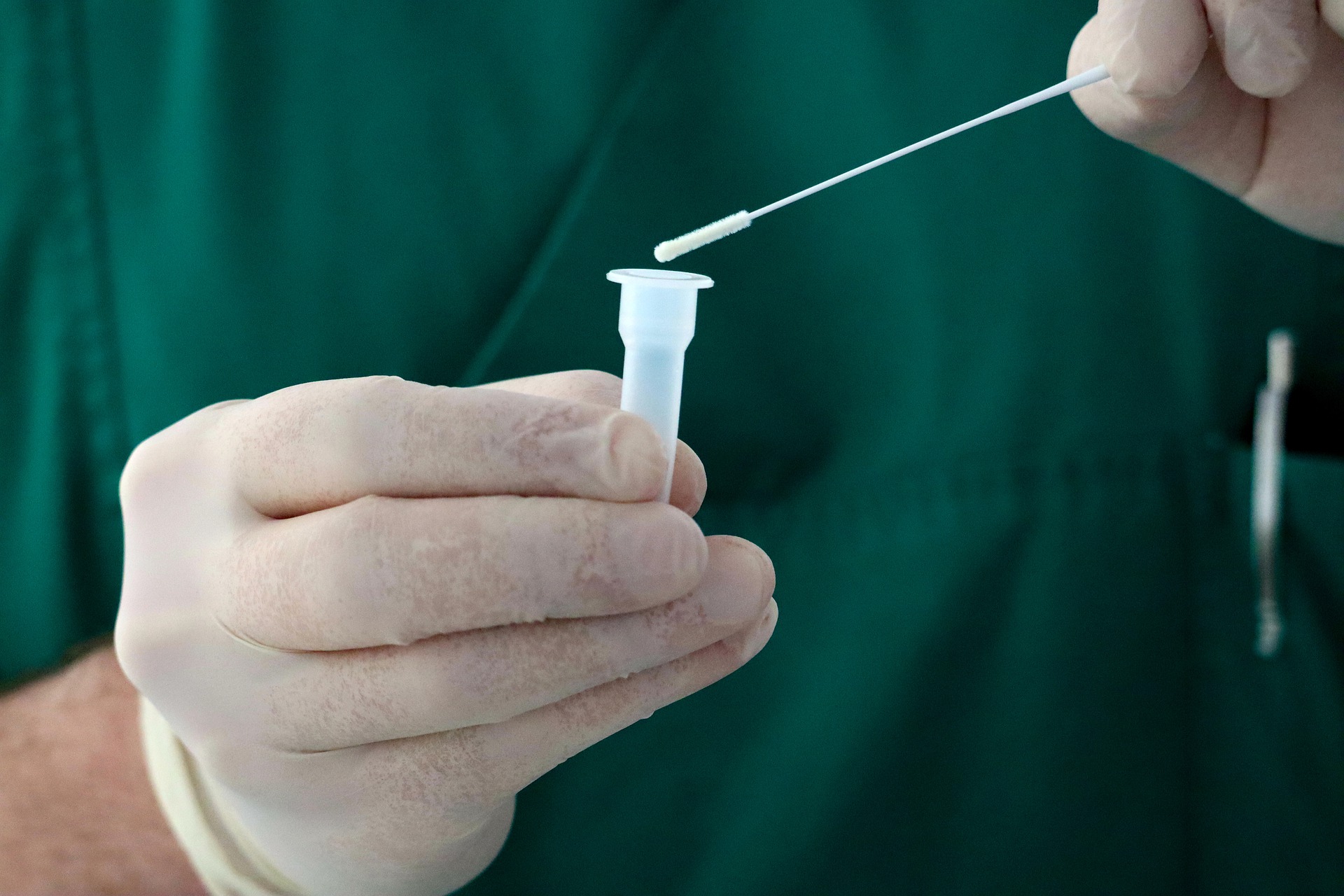
Genetic testing of saliva samples identifies the SARS-CoV-2 virus more quickly than testing of nasal swabs. The research is published March 21 in Microbiology Spectrum, a journal of the American Society for Microbiology.

In January 2021, Mindray Medical signed an AI cooperation framework agreement with Tencent AI Lab.
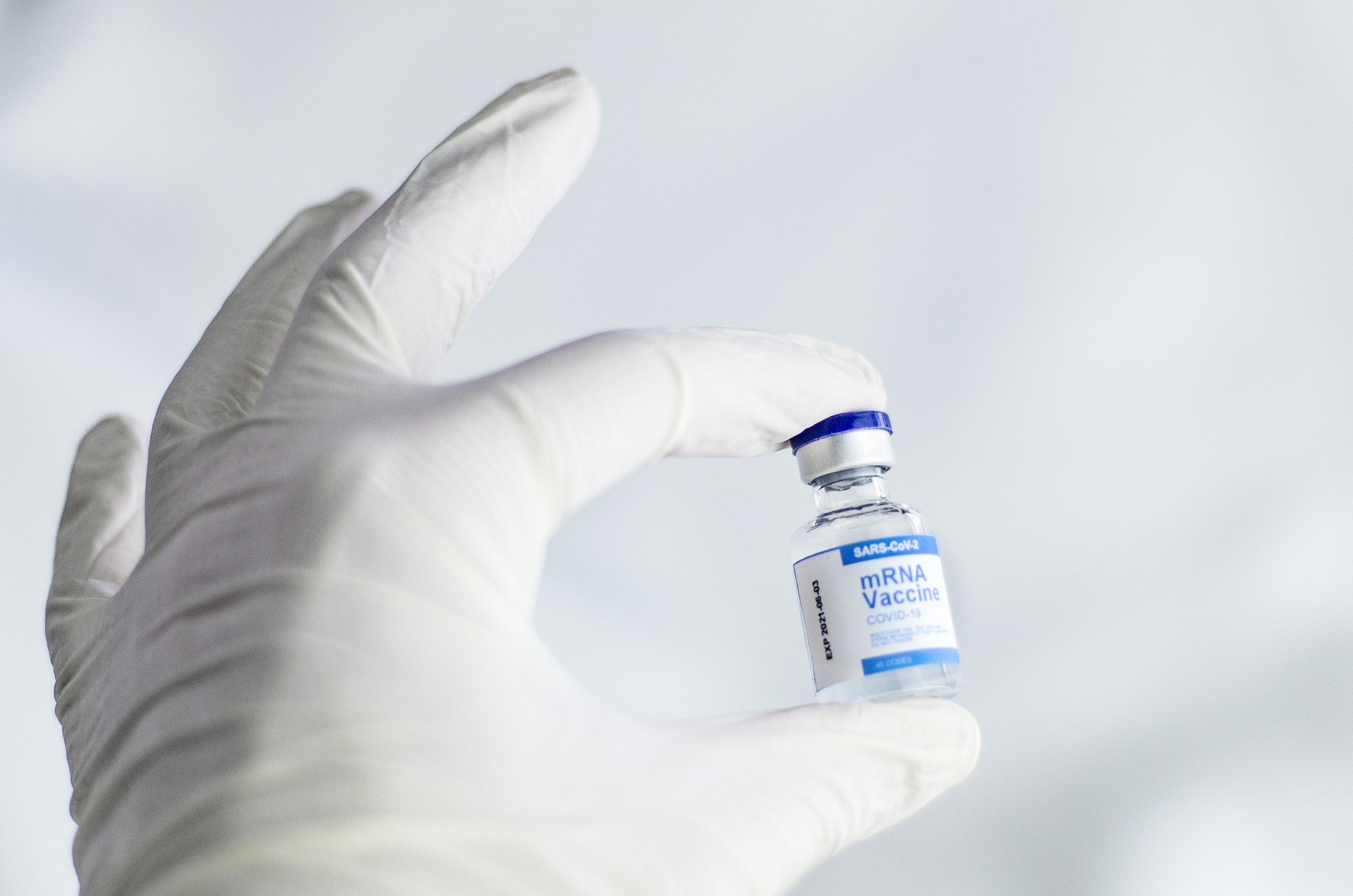
Moderna, Pfizer, and BioNTech are seeking FDA approval for a fourth dose of their respective COVID-19 vaccines.
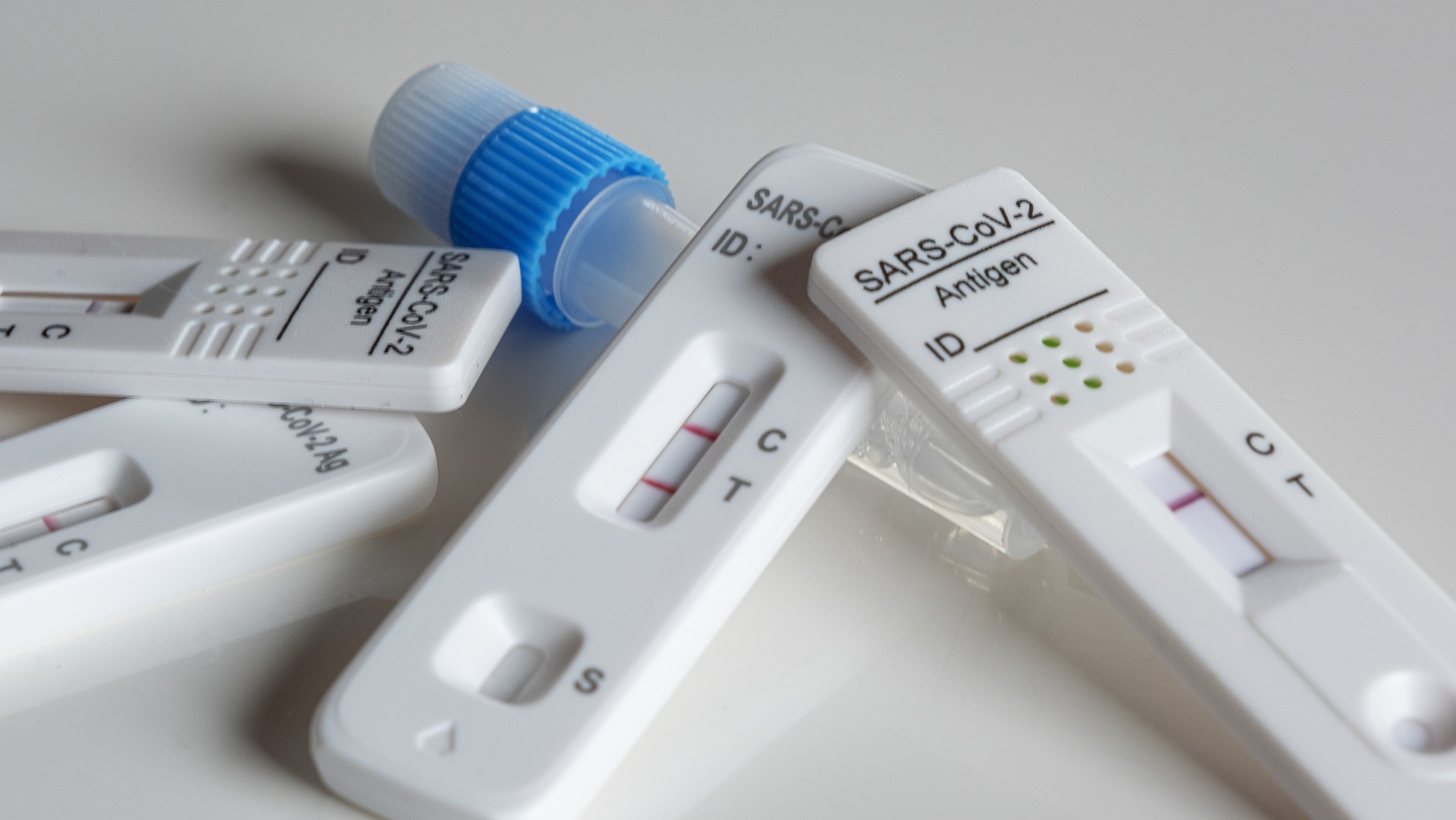
The US Food and Drug Administration last week granted Emergency Use Authorization for a SARS-CoV-2 molecular test developed by Helix.

According to the information of the official website China Food and Drug Administration, in March 18th, the 3 Shanghai COVID-19 antigen detection reagent products were approved.Up to now, China Food and Drug Administration has approved 17 COVID-19 antigen detection reagents.

BioMérieux said on Friday that it has received 510(k) clearance from the US Food and Drug Administration for its Vitek MS Prime MALDI-TOF mass spectrometry identification system.

The US Food and Drug Administration last week granted separate Emergency Use Authorizations for two SARS-CoV-2 antigen tests developed by Siemens Healthineers.
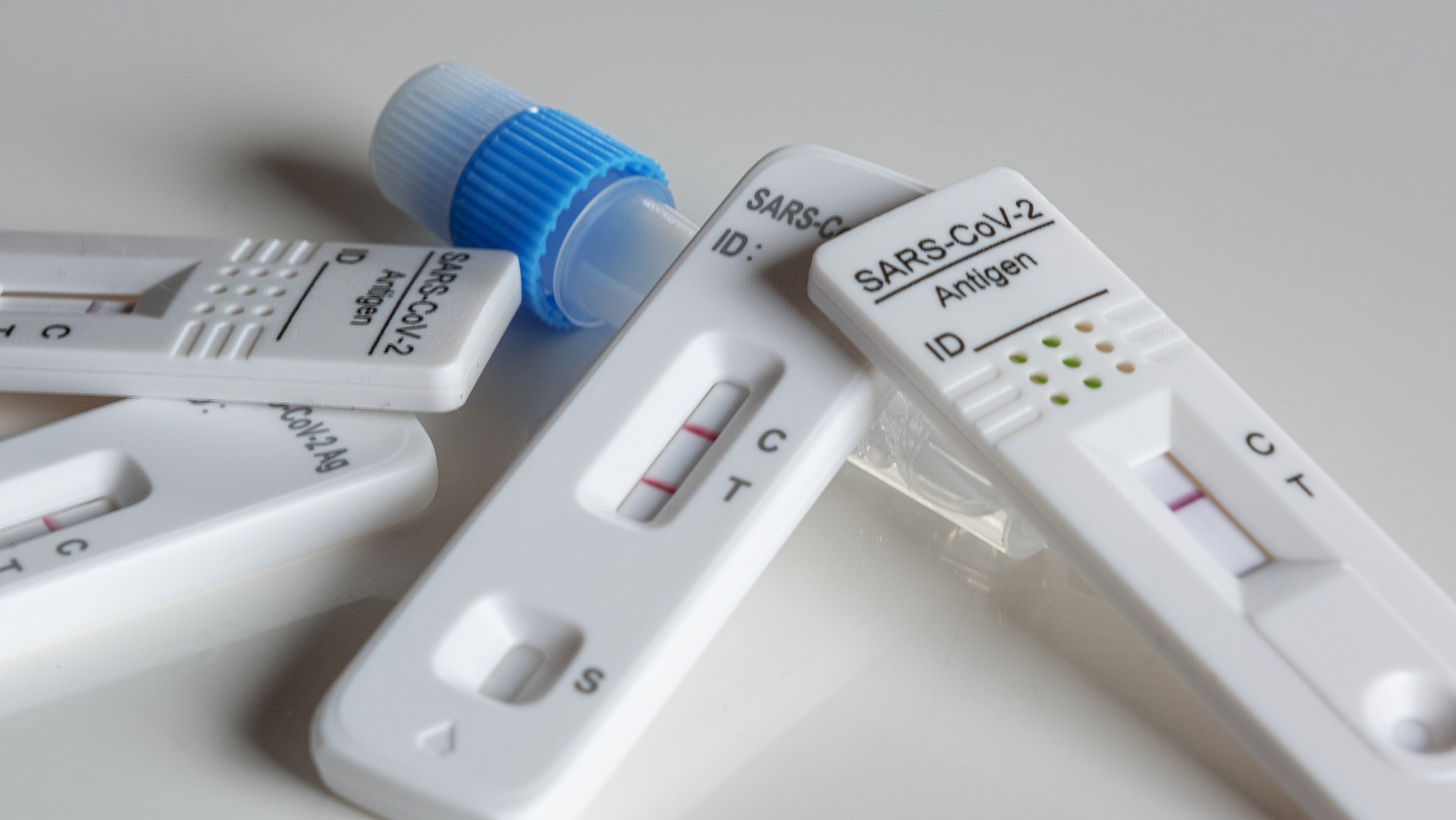
On March 17th, NMPA has approved the 14th New Coronavirus (2019-nCoV) antigen detection kit (colloidal gold method) produced by Zybio. Up to now, 14 COVID-19 antigen detection kits have been approved for sale.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
