

It can be seen from analysis on major blood grouping analyzer-related patent applications published in the 3 years from 2018 to 2021 that benefited from the basically stable original technologies of the industry; domestic enterprises have continuously increased R&D inputs, constantly improved technical innovation capability, made breakthroughs in many technologies, and developed a batch of core components of instruments with independent intellectual property rights through follow-on R&D strategy.

China plans to accelerate the establishment of a tiered healthcare delivery system, focusing on strengthening primary-level medical capacity to manage common and chronic diseases such as hypertension and diabetes, authorities said.

Becton Dickinson announced Wednesday that it has received US Food and Drug Administration approval for its automated, sample-to-answer, high-throughput BD Cor system along with the BD Onclarity HPV Assay for extended genotyping of human papillomavirus.

On August 20, Fosun Diagnostics, a holding subsidiary of Fosun Pharma, signed the investment agreement with Suzhou Abcarta. After the completion of the transaction, Fosun Diagnostics will hold about 58.67% equity of Suzhou Abcarta.

Genetron Health on Tuesday reported a 38 percent year-over-year increase in its second quarter revenues amid strong growth in sales from its LDT and IVD segments.

Cerba Research and ACT Genomics said on Tuesday that they have formed a joint venture to provide clinical trial services to clients throughout Asia Pacific through a new laboratory facility in Taiwan.
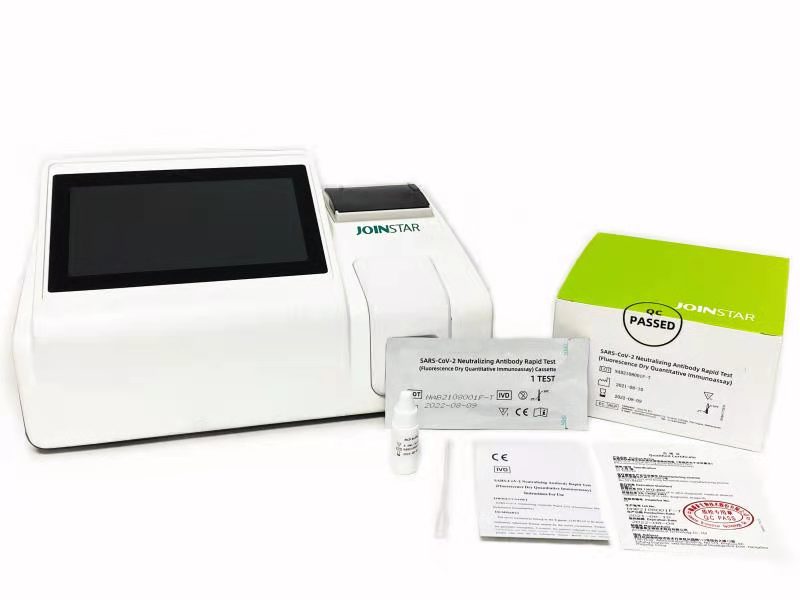
Recently, SARS-CoV-2 Neutralizing Antibody Rapid Test (Fluorescence Dry Quantitative Immunoassay) developed by Joinstar Biomedical Technology Co., Ltd. and Nanshan Pharmaceutical Research Institute of Guangdong province was officially sold to overseas markets.

On August 21, TELLGEN posted a 157.93% year-over-year increase on continued demand for its tumor and autoimmunity test.

AnPac Bio-Medical Science, a cancer screening and detection company, said on Monday that it has filed for registration testing of its Class III, multi-cancer detection device with China's National Medical Products Administration.

Point-of-care diagnostics firm LumiraDx and special purpose acquisition company CA Healthcare Acquisition said on Monday that they have revised the terms of their previously announced merger to lower the equity value of the combined company.
Waltham, MA-based Thermo Fisher Scientific was granted emergency use authorization for the TaqPath COVID-19 Fast PCR Combo Kit 2.0 and the TaqPath COVID-19 RNase P Combo Kit 2.0 assays.

The European Commission has opened an investigation assess whether Illumina's decision to complete its acquisition of Grail is a breach of the “standstill obligation”
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
