

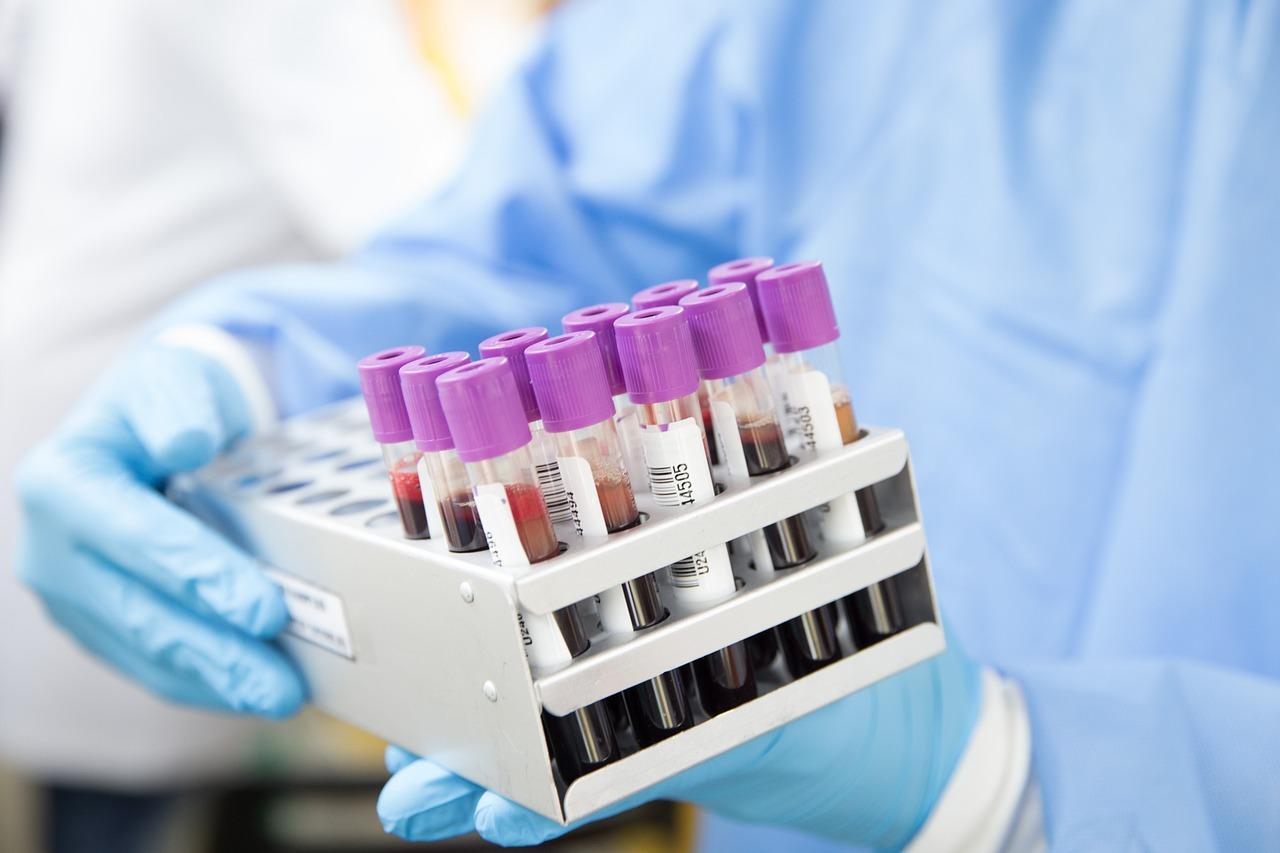
It can be seen from analysis on major blood grouping analyzer-related patent applications published in the 3 years from 2018 to 2021 that benefited from the basically stable original technologies of the industry; domestic enterprises have continuously increased R&D inputs, constantly improved technical innovation capability, made breakthroughs in many technologies, and developed a batch of core components of instruments with independent intellectual property rights through follow-on R&D strategy.

China plans to accelerate the establishment of a tiered healthcare delivery system, focusing on strengthening primary-level medical capacity to manage common and chronic diseases such as hypertension and diabetes, authorities said.

As the coronavirus continues to spread, companies are ramping up efforts to develop diagnostics. The latest diagnostic comes from CerTest Biotec and Franklin Lakes, NJ-based Becton Dickinson and Company (BD). The duo announced Tuesday the V

BillionToOne wants to expand its diagnostic offerings and its most recent financing could give it a push in that direction. The Menlo Park, CA-based company has raised $15 million in a follow-on to its series A round. With this financing, t

Released in conjunction with National Patient Safety Awareness week, ECRI InstitutesTop 10 Patient Safety Concerns for 2020raises the profile of safety issues that pose risks to patients and healthcare providers. The report relies on the an

FDA Halts Foreign Inspections In a rare action that is sure to impact medical device application reviews, FDA suspended inspections outside the United States through April in response to the coronavirus pandemic. The decision was based on g

Tests for the coronavirus (COVID-19) continue to roll in as the U.S. faces a public health crisis. Thermo Fisher Scientific and Mayo Clinic are the latest with diagnostic contributions. Waltham, MA-based Thermo Fisher is the second company

BD (Becton, Dickinson and Company) was one of several medtech companies that joined President Donald Trump when he declared a national emergency last week. Several medtech executives detailed their response to the novel coronavirus (COVID-1

Oncocyte said it has completed Clinical Laboratory Improvements Amendments (CLIA) validation for its DetermaIO. The Irvine, CA-based company said the diagnostic, which was previously known as the Insight Genetics IM Score Test, is used in l

FDA issued a letter to industry that includes steps the agencysCenter for Devices and Radiological Health (CDRH) has taken to prioritize work that advances the nations response during the Coronavirus (COVID-19) public health emergency. The

NEW YORK A new UK-based research consortium is betting that sequencing the virus that causes COVID-19 can provide policymakers with a clearer picture of the spread of the epidemic across the country as diagnostic testing remains limited. I

NEW YORK Pangolins may be an intermediary host for SARS-CoV-2, the virus causing the ongoing COVID-19 pandemic, a new sequencing analysis has found. While bats are thought to be the likely reservoir of SARS-CoV-2, experts suspect another ho
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
