


At present, MGT-based blood group test card has been used extensively in the field of blood typing, and MGT boasts of easy operation, high sensitivity, accurate results that can be saved and other strengthens, which allowed it to become method recommended by American Association of Blood Banks (AABB), technique recognized by FDA and internationally recognized blood group analysis test technique.

Stay up-to-date with the latest happenings in the rapidly evolving field of In Vitro Diagnostics (IVD) in China.

Foundation Medicine announced on Monday that it is expanding its partnership with Sequanta Technologies to provide its genomic profiling tests for research and oncology therapy development in China.

Switzerland's Roche (ROG.S) has agreed to pay an initial $7.1 billion to Roivant (ROIV.O) and Pfizer (PFE.N) for rights to a new inflammatory bowel disease drug, as it expands into new fields to offset falling oncology sales.

Roswell Park Cancer Center said this week that Agilent Technologies has licensed its blood cancer assay, PanHeme.
The World Health Organization on Thursday announced that it has published a fourth edition of its essential diagnostics list (EDL) adding tests for the hepatitis E virus and recommendations related to access to personal-use glucose monitoring devices.

Thermo Fisher Scientific and Boehringer Ingelheim said on Thursday that they have partnered to develop companion diagnostic tests to help identify patients with non-small cell lung cancer (NSCLC) with specific mutations who may benefit from emerging targeted therapies.

Group sales grow by 1% at constant exchange rates (CER) in the first nine months, showing a strong increase of 7% in the third quarter.

Oxford Nanopore Technologies PLC (LSE:ONT) has secured a £70 million investment from French in-vitro diagnostics (IVD) company bioMérieux, in exchange for a 3.5% stake.
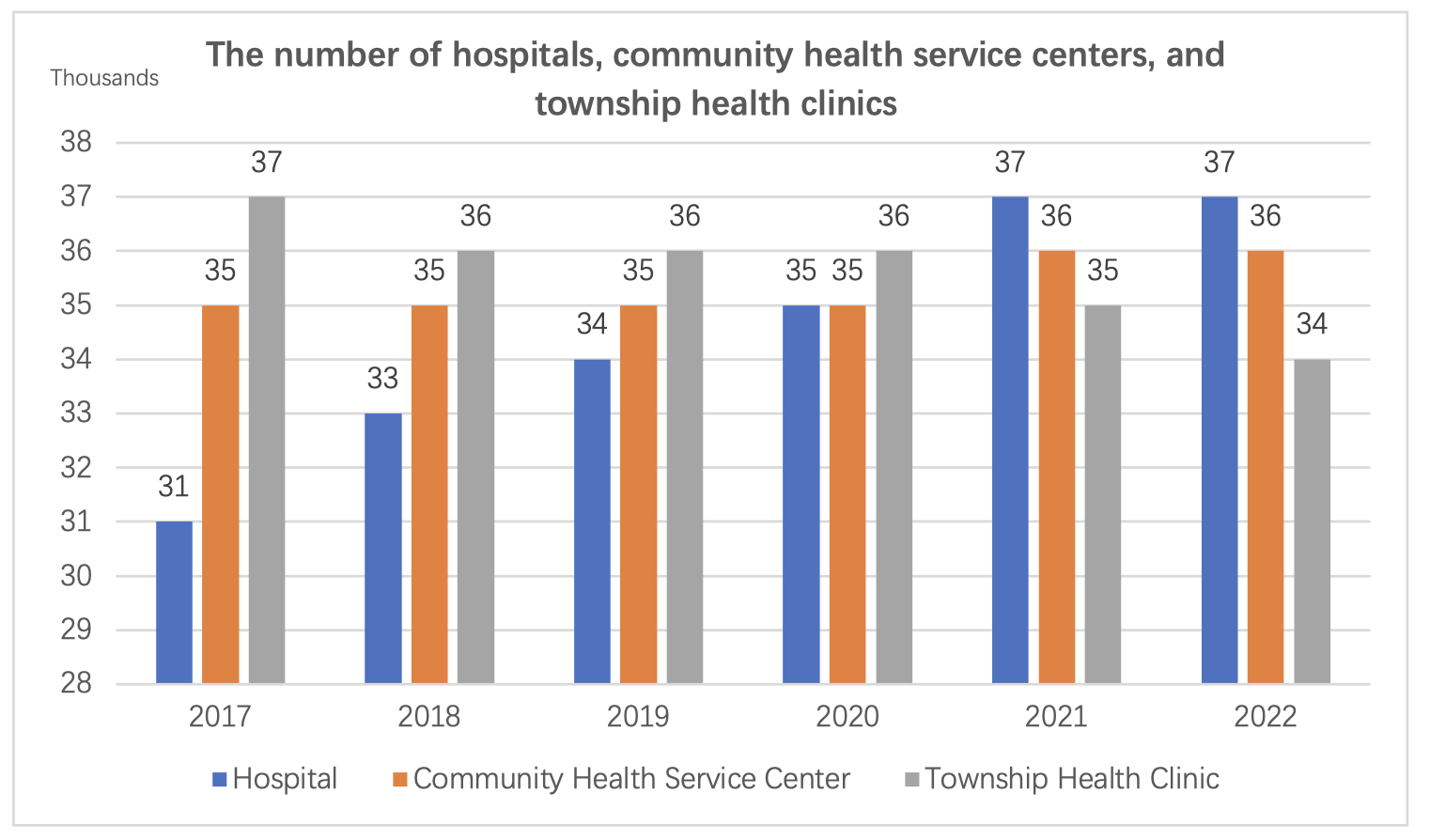
By the end of 2022, the total number of medical and health institutions nationwide reached 1,032,918, an increase of 1,983 compared to the previous year.

Revvity and Danaher subsidiary Sciex said Tuesday that they have signed a distribution agreement that will improve the accuracy of mass spectrometry-based disease screening in newborns by combining Sciex's mass spectrometry instruments and Revvity's reagents.

Abbott (NYSE: ABT) today announced financial results for the third quarter ended Sept. 30, 2023.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
