


After evaluation by the American Chemical Society (ACS) and INSPEC, the English scientific journal VIEW, PCEM and WILEY, has been evaluated by Content Selection & Advisory Board,CSAB and officially Indexed in Scopus.
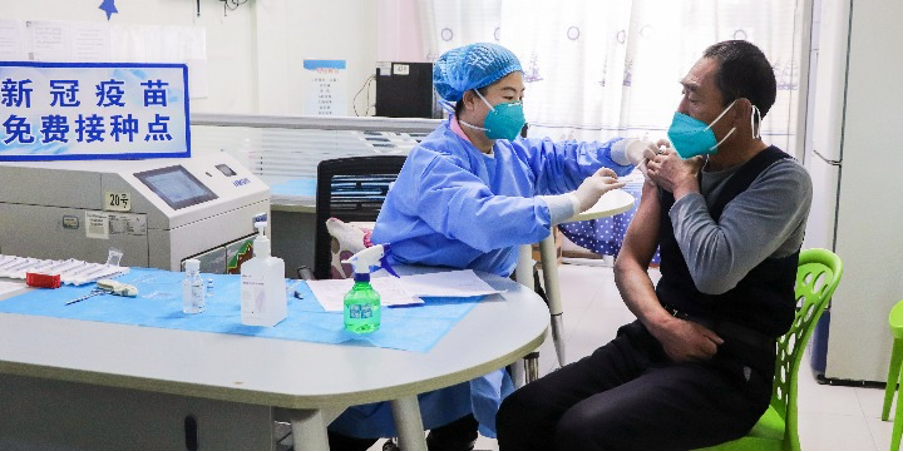
Editor's note: Major adjustments have been made to China's COVID-19 control policies, according to a 10-point notice released by the National Health Commission on Wednesday. Below are some of the key changes, to help you keep up to date.

Thermo Fisher Scientific said on Wednesday that its SeCore CDx HLA Sequencing System was granted de novo classification by the US Food and Drug Administration as a companion diagnostic to Immunocore’s Kimmtrak (tebentafusp-tebn) therapy for uveal melanoma.
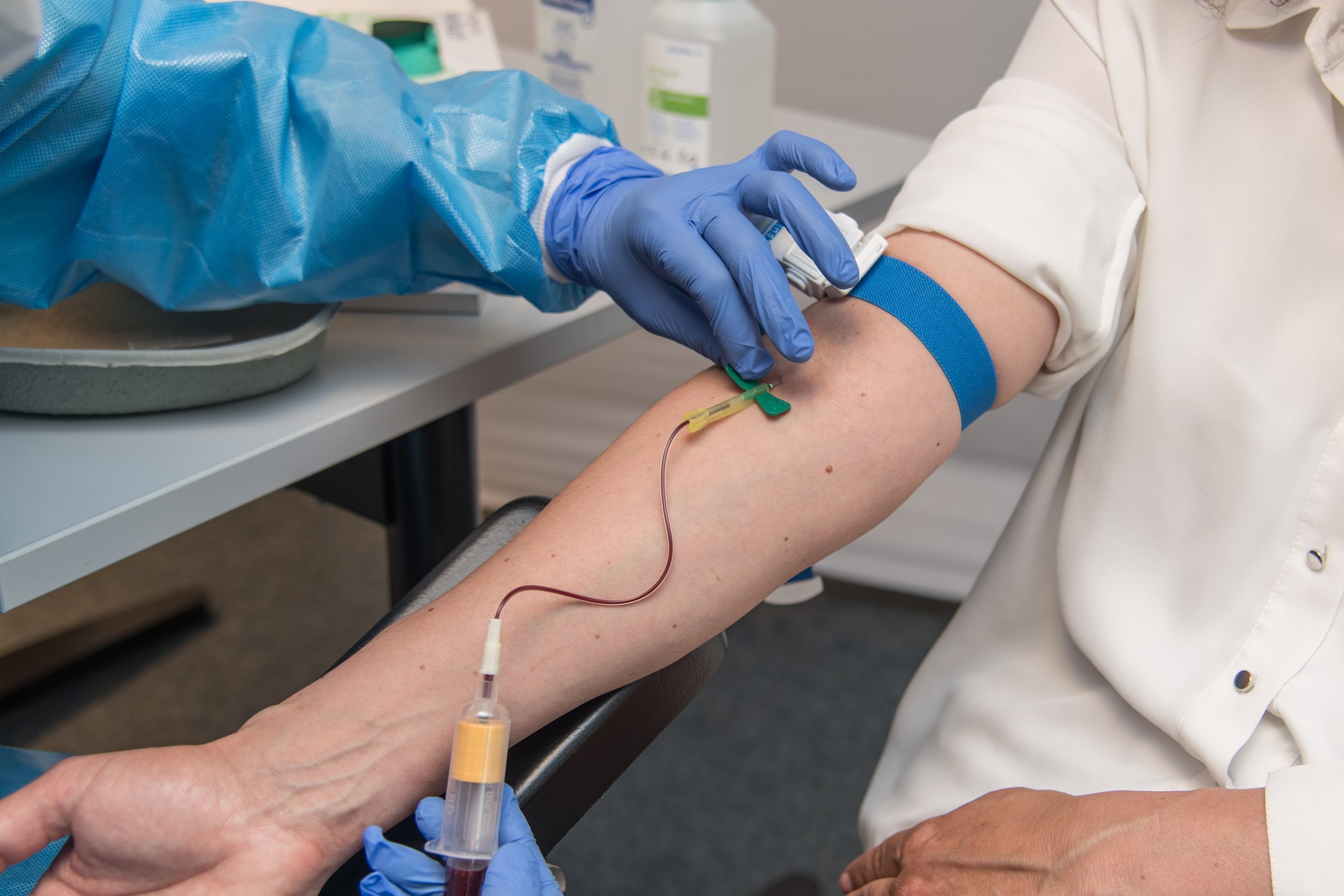
Scientists from the National University of Singapore (NUS) have discovered a novel low-cost method of testing for cancers. Called the Heatrich-BS assay, this new test sequences clinical samples that have been heated in order to isolate cancer-specific signatures found in a patient’s blood.

The European Commission has issued a Statement of Objections to Illumina and Grail on Monday, outlining the restorative measures it intends to adopt under the EU Merger Regulation.

Leica Biosystems has acquired Cell IDx, Inc., a leader in multiplexed tissue profiling. Founded in 2012 and headquartered in San Diego, California, Cell IDx provides multiplex staining panels, tissue staining, and imaging and analysis services.

Snibe reference testing laboratory passed all nine projects (seven in clinical enzymology, one in vitamins and one in hormones) in which it participated in the IFCC-RELA 2022.
According to CAIVD statistics, at present, the main in vitro diagnostic listed enterprises total: 62. They cover three segments: production, service and distribution. Among them, 46 are production enterprises, 11 are service enterprises and 5 are distribution enterprises.
A collaborative team led by researchers has re-engineered the process of microbial pathogen identification in blood samples from pediatric sepsis patients using the Wyss Institute's FcMBL broad-spectrum pathogen capture technology.

In addition, personnel, equipment, and facilities from the acquired companies will expand Reaction Biology’s presence in Germany while enabling its global customers to leverage Bioassay’s expansive portfolio of regulated clinical and commercial services.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
