
DiaCarta announced on Tuesday that its over-the-counter fecal occult blood test has received 510(k) clearance from the US Food and Drug Administration.

Foundation Medicine Inc. today announced that it has received approval from the U.S. Food and Drug Administration (FDA) for FoundationOne®CDx to be used as a companion diagnostic for AstraZeneca’s Truqap™ (capivasertib) in combination with Faslodex® (fulvestrant) which has been contemporaneously approved for the treatment of adult patients with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative locally advanced or metastatic breast cancer with one or more PIK3CA/AKT1/PTEN-alterations following progression on at least one endocrine-based regimen in the metastatic setting or recurrence on or within 12 months of completing adjuvant therapy.

The US Food and Drug Administration said Thursday that it has granted Roche Emergency Use Authorization for a new version of its multiplex molecular assay for detecting SARS-CoV-2 and influenza A/B.

The US Food and Drug Administration (FDA) has approved LetsGetChecked’s at-home testing system for chlamydia and gonorrhoea.

Agilent Technologies on Tuesday said it gained US Food and Drug Administration approval of the firm's PD-L1 companion diagnostic assay for use in identifying gastric or gastroesophageal junction adenocarcinoma patients for a combination treatment with Merck's Keytruda (pembrolizumab) checkpoint inhibitor.

Roche announced on Tuesday that its Cobas HPV test has received approval from the US Food and Drug Administration for use on the Cobas 5800 instrument.

Abbott announced on Thursday that its molecular test for human papillomavirus has received regulatory approval from the US Food and Drug Administration.

Werfen announced recently that its subsidiary Inova Diagnostics has secured US Food and Drug Administration 510(k) clearance for a blood-based immunoassay reagent used to aid the diagnosis of connective tissue diseases.

Abbott's RealTime IDH1 Assay has received approval from the US Food and Drug Administration as a companion diagnostic for Servier Pharmaceuticals' Tibsovo (ivosidenib) in patients with relapsed or refractory myelodysplastic syndrome, the FDA announced on Tuesday.
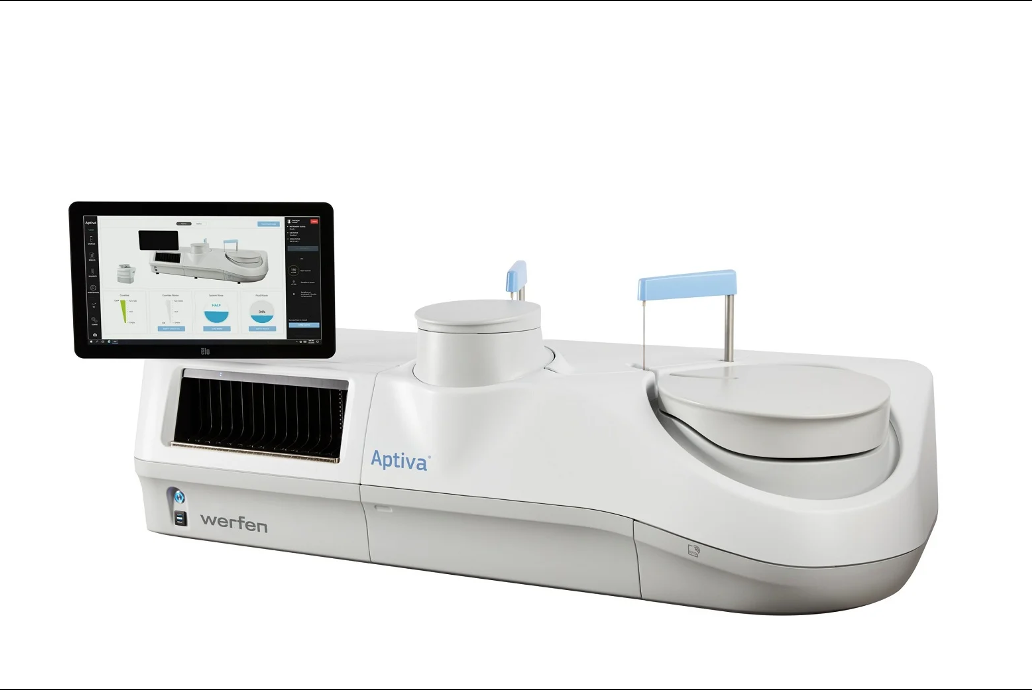
Werfen has received 510(k) clearance from the US Food and Drug Administration (FDA) for its Aptiva Connective Tissue Disease (CTD) Essential reagent.
✔ All (518)
✔ Press release (3)
✔ Industry news (515)
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
