
Roche subsidiary Foundation Medicine announced on Thursday that the US Food and Drug Administration has approved two of its companion diagnostic tests for use with Pfizer's Braftovi (encorafenib) in combination with Mektovi (binimetinib) for patients with metastatic non-small cell lung cancer with a BRAF V600E mutation.

As more and more medical technologies are being built on artificial intelligence and machine learning, the FDA is enlisting a new panel of outside experts to help it get deeper into the weeds.

Invitae (NYSE: NVTA), a leading medical genetics company, today announced it gained FDA market authorization for its Common Hereditary Cancers Panel.

Foundation Medicine Inc. today announced that it has received approval from the U.S. Food and Drug Administration (FDA) for FoundationOne®CDx to be used as a companion diagnostic for Eli Lilly and Company’s Retevmo (selpercatinib), which was approved by the FDA for the treatment of adult patients with locally advanced or metastatic solid tumors with a RET gene fusion that have progressed on or following prior systemic treatment or who have no satisfactory alternative treatment options.
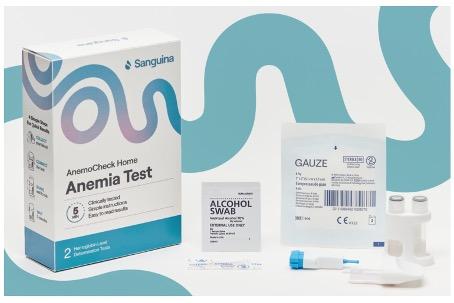
Today, Sanguina, Inc. (Sanguina), a leading biotech company, is pleased to announce the FDA clearance of AnemoCheck Home, the only FDA-cleared home hemoglobin test kit available in the United States.

Thermo Fisher Scientific said on Wednesday that it has obtained US Food and Drug Administration clearance for its BRAHMS CgA II KRYPTOR test, a chromogranin A (CgA) immunoassay for assessing tumor progression in gastroenteropancreatic neuroendocrine tumor (GEP-NET) patients.

Through MDUFA V, the FDA committed to several international harmonization initiatives and secured additional resources to support the work. The publication of a draft strategic plan was the first item on the MDUFA V convergence and reliance agenda.

Streck announced on Monday that its MDx-Chex quality controls have received clearance from the US Food and Drug Administration.

QuidelOrtho Corporation (Nasdaq: QDEL) (“QuidelOrtho”), a global provider of innovative in vitro diagnostic technologies designed for point-of-care settings, clinical labs and transfusion medicine, announced today that it has been granted CLIA Waiver from the U.S. Food and Drug Administration (FDA), applying to its new Sofia® 2 SARS Antigen+ FIA.

T2 Biosystems, Inc. (NASDAQ:TTOO), a leader in the rapid detection of sepsis-causing pathogens and antibiotic resistance genes, today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the T2Biothreat™ Panel. The FDA 510(k) clearance allows the company to immediately begin marketing and selling the T2Biothreat Panel in the U.S. market.
✔ All (518)
✔ Press release (3)
✔ Industry news (515)
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
