
Co-Diagnostics, Inc. (Nasdaq: CODX) ("Co-Dx," or "the Company"), a molecular diagnostics company with a unique, patented platform for the development of molecular diagnostic tests, today announced its financial results for the first quarter ended March 31, 2026.

Co-Diagnostics, Inc. (NASDAQ: CODX) ("Co-Diagnostics," "Co-Dx," or "the Company"), a molecular diagnostics company with a unique, patented platform for the development of molecular diagnostic tests, today announced its financial results for the full year ended December 31, 2025.

The World Health Organization (WHO) is issuing, for the first time, recommendations on new near-point-of-care (NPOC) molecular tests for the diagnosis of tuberculosis (TB); easy-to-collect tongue swab samples to simplify and expand access to testing; and a cost-saving sputum pooling strategy to increase testing efficiency for TB and rifampicin-resistant TB.

BIOMAKERS, a precision medicine and oncology intelligence company headquartered in San Francisco, today announced the closing of an additional $8 million. The financing marks a key inflection point as the company transitions from building a differentiated global diagnostics and data infrastructure to scaling an AI-native platform – while continuing to expand its comprehensive molecular testing reach across Latin America – designed to accelerate drug and diagnostic development, improve clinical trial execution, and enable precision oncology at global scale.

BillionToOne, Inc. (NASDAQ: BLLN), a next-generation molecular diagnostics company with a mission to create powerful and accurate tests that are accessible to all, today announced a collaboration with Epic, the nation's most widely used comprehensive electronic health record (EHR). The agreement, signed in December 2025, will integrate BillionToOne's prenatal and oncology testing portfolio with Epic's Aura diagnostics suite.

Synthego, a recognized leader in CRISPR solutions, announces its formal entry into the molecular and clinical diagnostic reagents sector. Building upon its reputation for rigorous quality, custom manufacturing, and innovation, Synthego's significant expansion of its product portfolio empowers researchers, clinicians, and diagnostic laboratories with high-quality, reliable reagents designed to accelerate the development and deployment of cutting-edge diagnostic tests.
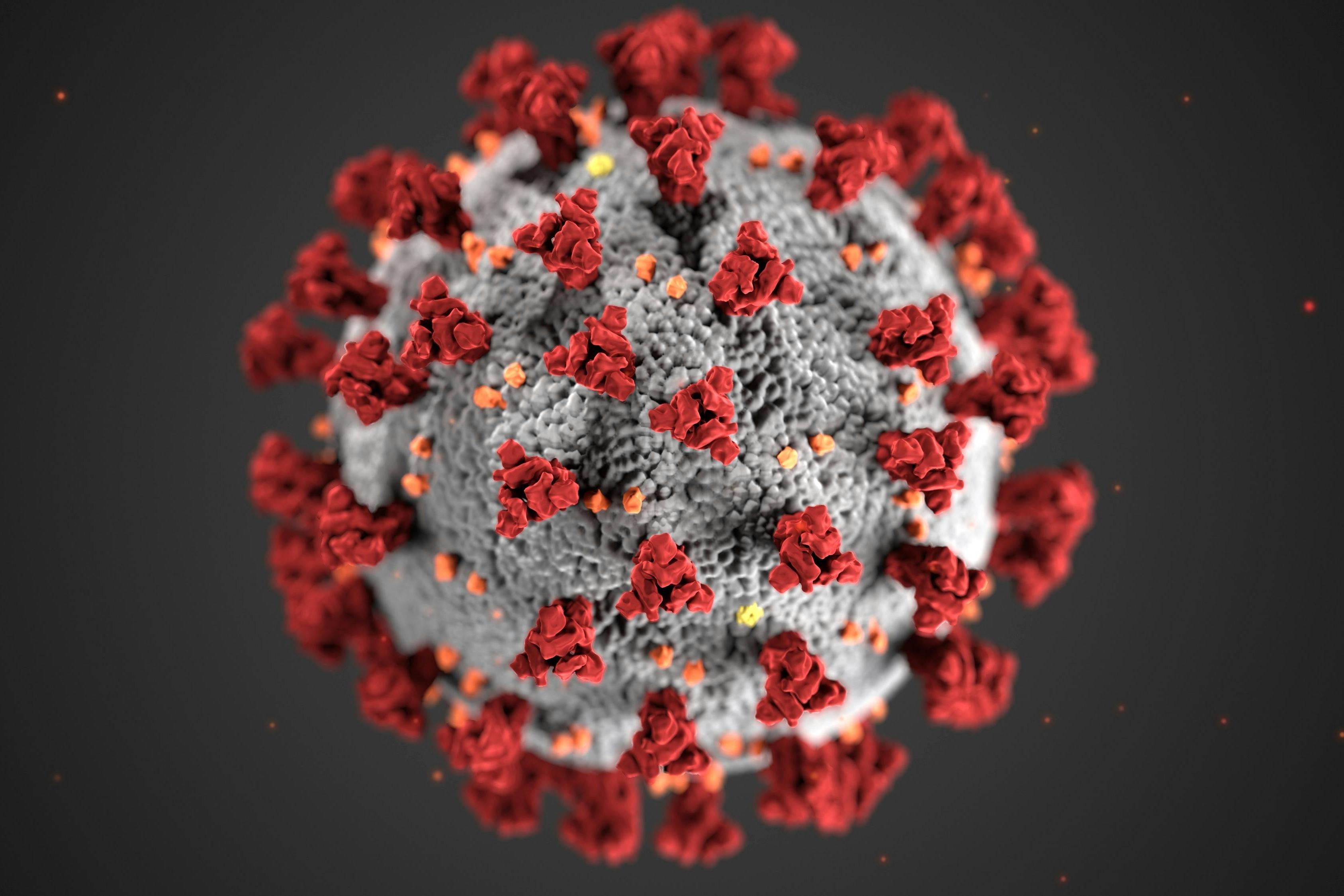
Following decades of cumulative efforts, a number of infectious disease tests and technologies broke new ground in 2025, hinting at rapid advances in the development of new systems and assays. But the past year also saw its fair share of upheaval, possibly suggesting more may be in store in 2026.

Chinese molecular diagnostics and precision medicine firm GenePlus Technology said on Sunday that it has filed for an initial public offering on the Hong Kong Stock Exchange.

Molecular diagnostics firm BillionToOne reported after the close of the market on Tuesday that its Q3 2025 revenues more than doubled year over year.

The Global Health Innovative Technology (GHIT) Fund said Monday that it has invested ¥70 million ($460,000) in the development of a prototype molecular assay to detect mpox.
✔ All (147)
✔ Press release (0)
✔ Industry news (147)
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
