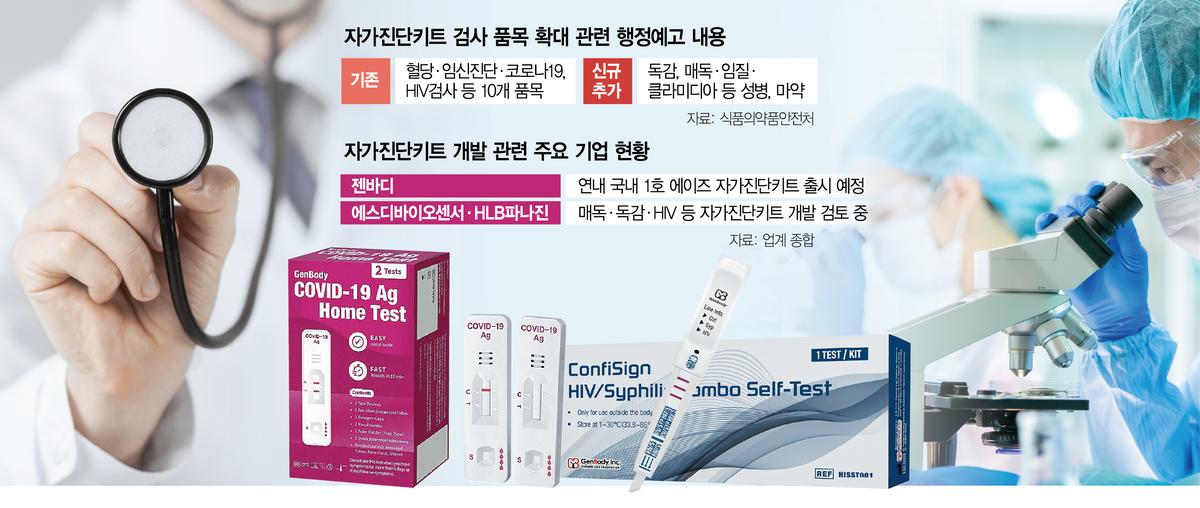
Korea's diagnostics industry is buzzing as the Ministry of Food and Drug Safety (MFDS) moves to expand the range of conditions that can be tested with home diagnostic kits to include influenza and sexually transmitted diseases. Market growth expectations are rising as diagnostic kits previously restricted to professional use become available to general consumers. Companies are accelerating development of self-testing kits in response to the policy shift.

Amoy Diagnostics Co., Ltd., Medical & Biological Laboratories Co., Ltd., a group company of Tokuyama Corporation, and Precision Medicine Asia Co., Ltd. today announced that MBL received approval from the Japanese Ministry of Health, Labour and Welfare (MHLW) on March 5, 2026 for the AmoyDx® PIK3CA Mutation Detection Kit in Japan. The approval in Japan marks an important milestone achieved through the close collaboration of MBL, AmoyDx and PREMIA.

Digital health company Biostarks has announced new at-home longevity test kits that leverage the firm’s mass-spectrometry platform.

Cue Health, a healthcare technology company, announced the nationwide launch of a new suite of consumer-friendly, at-home diagnostic test kits.

The US Food and Drug Administration in January modified existing 510(k) clearances for several firms, including Becton Dickinson and Roche, while it granted new clearances for diagnostic instruments and assays from Medica, Selux Diagnostics, and others, according to the agency website.

BGI Genomics released the 2022 annual fiscal forecast. The operating revenue is expected to reach CNY 6.4 to 7.4 billion; the net profit attributable to shareholders of the listed companies is forecast to be CNY 800 million to 1.04 billion.

On December 14, 2022, Autobio and Pushkang signed a strategic cooperation agreement in Zhengzhou, and the two sides will again open in-depth cooperation in the field of blood coagulation testing.

BGI Genomics said on Thursday that it has received CE-IVD marking for its next-generation sequencing-based test for thalassemia mutation detection.

On July 27, Illumina and KingMed Diagnostics Group Co., Ltd. reached a new strategic cooperation on tumor gene detection.
Recently, the Wondfo Biotech COVID-19 Antigen Detection Kit (Immunochromatography Method) was included in the WHO (World Health Organization) list for emergency use by the WHO, which can be purchased by other countries and regions.
✔ All (34)
✔ Press release (0)
✔ Industry news (34)
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
