
China plans to accelerate the establishment of a tiered healthcare delivery system, focusing on strengthening primary-level medical capacity to manage common and chronic diseases such as hypertension and diabetes, authorities said.

OSR Holdings, Inc. (NASDAQ: OSRH) today announced that it has executed a definitive agreement to acquire Woori IO Co., Ltd. ("WORIO"), a pioneer in noninvasive glucose monitoring (NIGM), via a comprehensive share exchange. The acquisition reinforces OSRH's dedication to advancing biomedical innovation by adding a potential breakthrough technology for diabetes care, bringing truly needle-free glucose monitoring closer to patients globally and promising a safer, more convenient and accurate alternative to currently available technologies.

Revvity, Inc. (NYSE: RVTY) today announced a program for expansion of its type 1 diabetes (T1D) offering to include a population-scale assay for early detection with support from Sanofi (EURONEXT: SAN and NASDAQ: SNY). Revvity will develop a T1D 4-plex in vitro diagnostic (IVD) assay based on its existing research-use (RUO) 3-plex assay.

Renalytix plc (LSE: RENX) (OTCQB: RNLXY), today announced an agreement with Tempus AI, Inc. (NASDAQ: TEM) to make kidneyintelX.dkd prognostic blood testing more widely available for eligible patients within its US network of healthcare institutions. Eligible patients have type 2 diabetes with chronic kidney disease, impacting nearly 15 million individuals in the US.

Medtronic plc (NYSE: MDT), a global leader in healthcare technology, today announced financial results for its first quarter (Q1) of fiscal year 2026 (FY26), which ended July 25, 2025.

Medtronic plc (NYSE: MDT), a global leader in healthcare technology, today announced financial results for its fourth quarter (Q4) and fiscal year 2025 (FY25), which ended April 25, 2025.

By 2030, a diabetes prevention and treatment system will enable over 60% of residents aged 18 and above to be aware of the disease. By then, the coverage rate of standardized management services for type 2 diabetes patients at the primary level will exceed 70%, according to the documents released by multiple departments including the National Health Commission (NHC).

Abbott (NYSE: ABT) today announced U.S. Food and Drug Administration (FDA) clearance for two new over-the-counter continuous glucose monitoring (CGM) systems – Lingo™ and Libre Rio™, which are based on Abbott's world-leading FreeStyle Libre® continuous glucose monitoring technology1, now used by about 6 million people globally2. The newly cleared systems have been intentionally designed to meet different needs – Lingo for consumers who want to better understand and improve their health and wellness, and Libre Rio for adults with Type 2 diabetes who do not use insulin and typically manage their diabetes through lifestyle modifications.

An analysis conducted by an international research team has identified 13 biomarkers that may improve the accuracy of predicting the risk of cardiovascular disease (CVD) in people with type 2 diabetes.
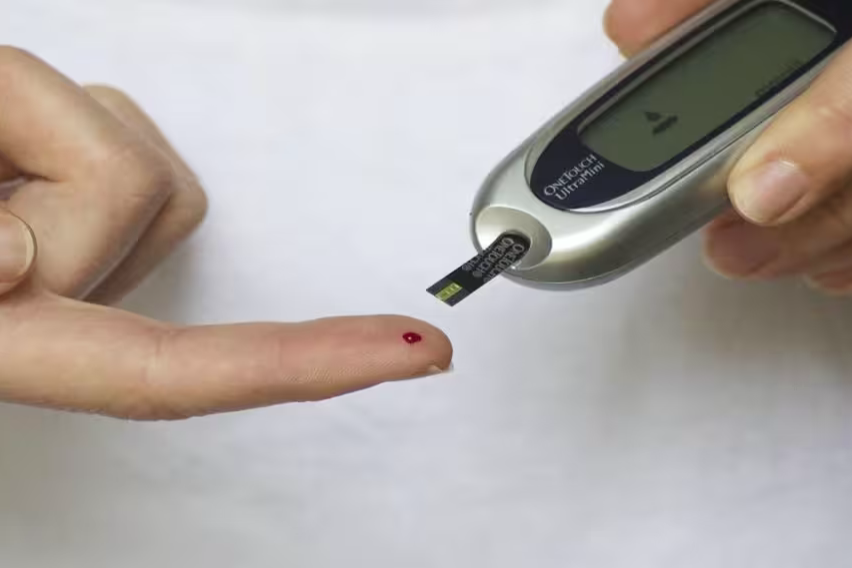
Roche Holding AG will fold its stand-alone diabetes business into its broader diagnostics unit after a decade and a half of declining sales.
✔ All (22)
✔ Press release (0)
✔ Industry news (22)
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
