
Abbott (ABT.N), opens new tab cut its annual profit forecast on Thursday due to an impact from its $23 billion acquisition of cancer test maker Exact Sciences, sending its shares down 5% even as the medical device maker narrowly beat quarterly results estimates.
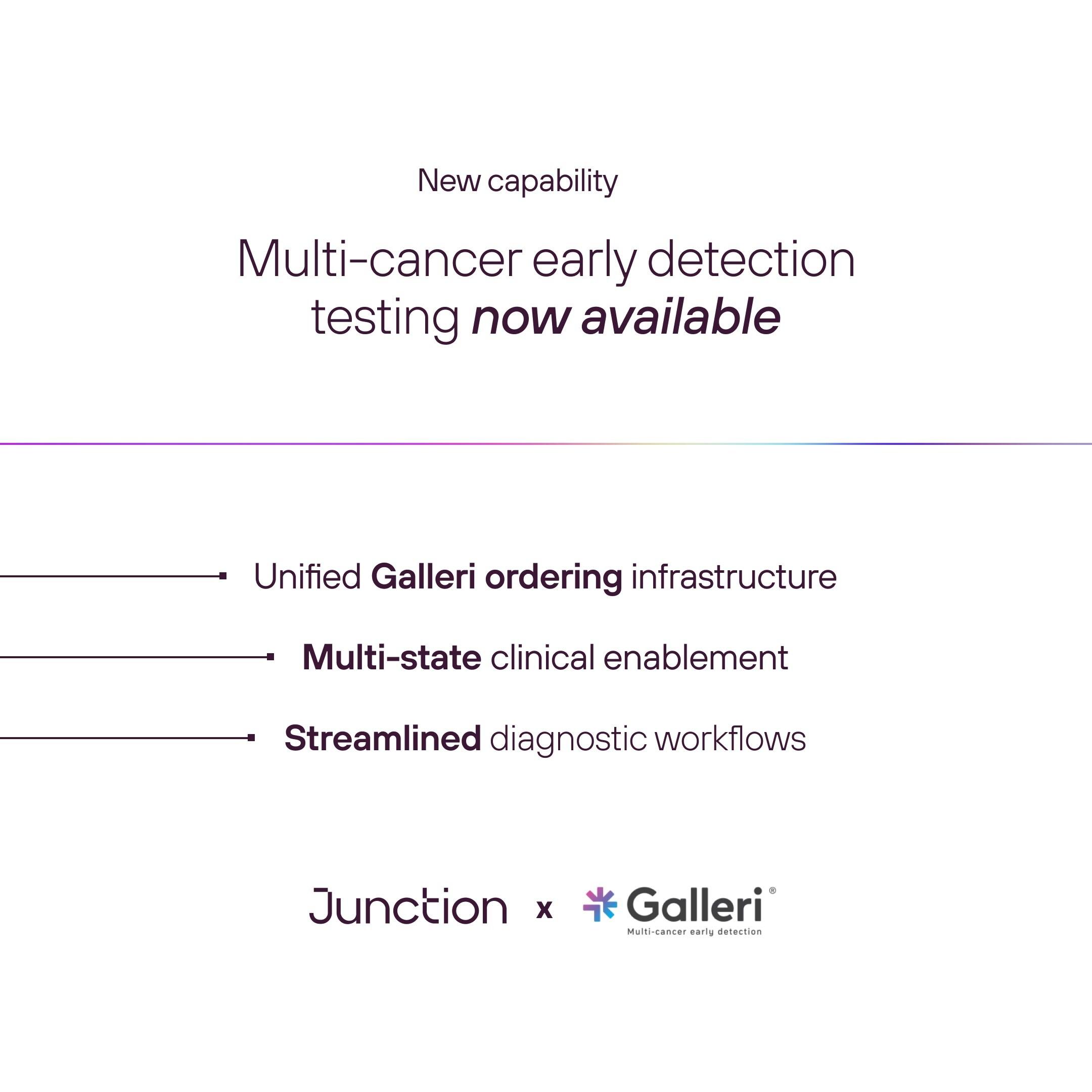
Junction, the infrastructure platform for diagnostic testing and health data workflows, today announced a collaboration with GRAIL to support scalable access to the Galleri® multi-cancer early detection test across digital health platforms and clinical organizations.

Merck (MRK.N), opens new tab said on Monday it would create a separate division for its cancer business, anchored by its top-selling drug Keytruda, as the U.S. drugmaker braces for the upcoming patent expiry of the blockbuster treatment.

US pharmaceutical group Merck is in talks to buy Revolution Medicines, a cancer drugmaker with a market capitalisation of more than $20bn in what would be the latest big deal in the red-hot biotechnology sector.

SOPHiA GENETICS (NASDAQ: SOPH), a global leader in AI-driven precision medicine, and The University of Texas MD Anderson Cancer Center today announced a strategic collaboration that unites SOPHiA GENETICS' AI-powered analytics with MD Anderson's clinical and scientific expertise to accelerate data-driven cancer care through new tools that can accurately analyze, interpret and translate diagnostic results into clinical practice.

Chinese molecular diagnostics and precision medicine firm GenePlus Technology said on Sunday that it has filed for an initial public offering on the Hong Kong Stock Exchange.

Singlera Genomics said on Tuesday that it has entered into a research and distribution agreement with EU-based international medical company Pure Medical for its cell-free DNA detection products.

Zydus Lifesciences said on Friday that it has signed an agreement to exclusively commercialize a trio of Myriad Genetics cancer tests in India.

Guardant Health Japan said Monday that it has received approval from that country's Ministry of Health, Labour, and Welfare (MHLW) for its Guardant360 CDx as a companion diagnostic for identifying ESR1 mutations in breast cancer patients.

Results of a Phase I clinical trial published on Tuesday demonstrate that Personalis' minimal residual disease (MRD) assay, NeXT Personal, accurately predicts immunotherapy response across multiple metastatic solid tumor cancer types.
✔ All (183)
✔ Press release (1)
✔ Industry news (182)
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
