
Artera, a developer of multimodal AI-based prognostic and predictive cancer tests, announced today that it was granted CE marking under the European Union In Vitro Diagnostic Regulation (EU IVDR) for both its ArteraAI Prostate Biopsy Assay and ArteraAI Breast Cancer Assay, marking a major step in the company’s global expansion.

Myriad Genetics Inc., (NASDAQ: MYGN), a leader in molecular diagnostic testing and precision medicine, today announced that Japan’s Ministry of Health, Labour and Welfare (MHLW) has approved the use of the MyChoice® Test for prostate cancer patients as a companion diagnostic for Lynparza® (olaparib). With this approval, clinicians can now order the MyChoice Test to determine homologous recombination deficiency (HRD) status for patients with ovarian cancer, and BRCA1/2 status for breast and prostate cancers.

Mdxhealth and the University of Oxford will test whether the firm's Genomic Prostate Score (GPS) assay can predict disease progression and treatment outcomes in prostate cancer patients, the company said Tuesday.
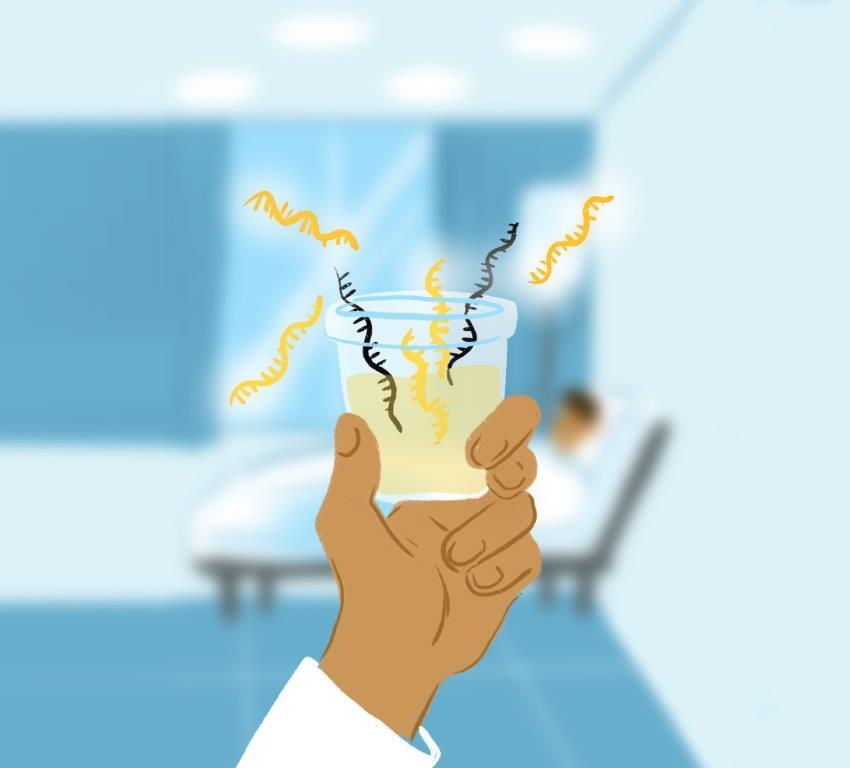
Researchers at the Johns Hopkins Kimmel Cancer Center, Johns Hopkins All Children’s Hospital and four other institutions have devised a novel method to test for prostate cancer using biomarkers present in urine, funded in part by the National Institutes of Health. This approach could significantly reduce the need for invasive, often painful biopsies, they say.

Artera, the developer of multimodal artificial intelligence (MMAI)-based prognostic and predictive cancer tests, announced today that the U.S. Food and Drug Administration (FDA) has granted De Novo authorization for ArteraAI Prostate, establishing it as the first and only AI-powered software authorized to prognosticate long-term outcomes for patients with non-metastatic prostate cancer.

Veracyte, Inc. (Nasdaq: VCYT), a leading cancer diagnostics company, today announced that its market-leading Decipher Prostate Genomic Classifier is the only gene expression test to be included in version 1 of the 2025 NCCN* Clinical Practice Guidelines in Oncology (NCCN Guidelines®) as part of the updated “Advanced Tools” table located in the Principles of Risk Stratification and Biomarkers section (PROS-H) .

Trinity Biotech plc (Nasdaq: TRIB), a commercial-stage biotechnology company focused on human diagnostics and diabetes management solutions, including wearable biosensors, today announced the acquisition of EpiCapture Limited, a company developing a non-invasive test for monitoring the risk of aggressive prostate cancer. This acquisition marks Trinity Biotech’s strategic expansion into the oncology diagnostics market.

DiaCarta, Inc. ("DiaCarta"), a pioneer in molecular diagnostic test development for cancer and infectious diseases, today announced that it has established a strategic collaboration with OncoAssure Ltd, an Irish medical diagnostics company. The focus of the collaboration is to commercialize OncoAssure's groundbreaking Prostate test which is designed to identify patients with a lower risk of prostate cancer recurrence, guiding decisions on active surveillance or reduced monitoring post-treatment.

Digital pathology firm Paige said Thursday it has forged a partnership with Ohio State University's Wexner Medical Center for a study to evaluate the use of artificial intelligence-assisted tools to diagnose prostate cancer in a community hospital setting.

Roche subsidiary Foundation Medicine announced on Monday that its FoundationOne CDx test received approval from the US Food and Drug Administration for use as a companion diagnostic for Janssen Biotech's Akeega (niraparib and abiraterone acetate).
✔ All (13)
✔ Press release (0)
✔ Industry news (13)
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
