


Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Themed "Global IVD Innovation & Cross-Regional Integration," this groundbreaking event brings together distinguished experts from around the world to explore the future of diagnostics.

The US Food and Drug Administration this week granted separate Emergency Use Authorizations for a respiratory panel RT-PCR test from Laboratory Corporation of America and a COVID-19 RT-PCR test from Nexus Medical Labs.
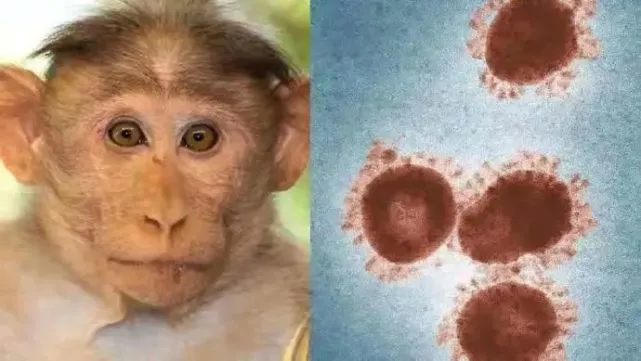
The WHO office in Congo (DRC) released a message on May 20, local time, saying that more than 1,200 people suspected of being infected with monkeypox virus. Virus spread across 18 provinces across the country and caused 58 people died.

6 COVID-19 test kits obtained CE mark

Cepheid announced on Thursday that it has obtained the CE mark for a rapid molecular test to detect the SARS-CoV-2 virus. The new test will begin shipping this month.

LumiraDx on Thursday announced it has obtained CE marking for its five-minute SARS-CoV-2 Ag Ultra Test.

On May 18, the treatment report of the first Chinese produced SARS-CoV-2 oral drug was announced.

The Food and Drug Administration published a Class I recall report for two COVID-19 testing products that covers the removal of more than 51,000 unauthorized coronaviurs tests from the U.S. market.

Genomics and diagnostics company Prenetics ended its first day as publicly traded company on the Nasdaq on Wednesday with its stock down 9 percent.

On May 18, Yunkang Group was listed on Hong Kong Exchanges and Clearing Limited (HKEX), stock code: 2325.HK. At press time, it opened down over 10% with a total market capitalisation of approximately HK$4.3 billion.

On Monday, The US Food and Drug Administration issued an Emergency Use Authorization to Laboratory Corporation of America's Seasonal Respiratory Virus RT-PCR DTC test for use without a prescription by individuals with symptoms of COVID-19.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
