Original from: Medical Device Network
Werfen has received CE marking for its Aptiva antiphospholipid syndrome (APS) Immunoglobulin G (IgG) and Immunoglobulin M (IgM) reagents, meaning the in vitro diagnostics can now be used in the EU.
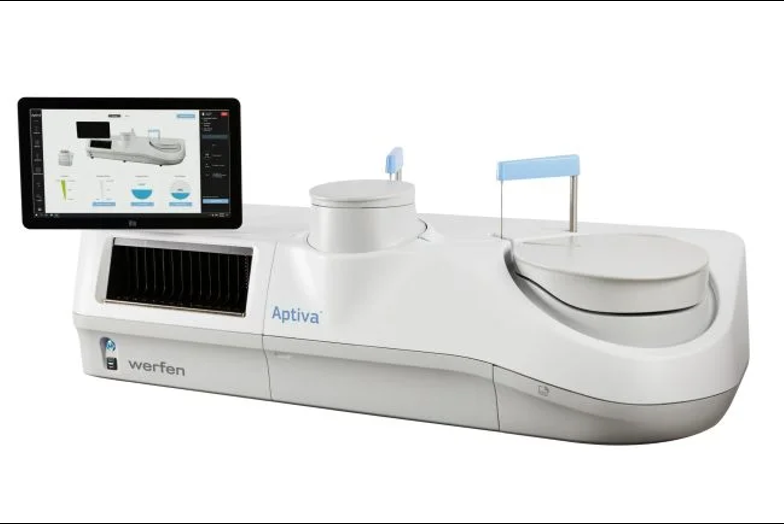
The reagents are immunoassays that use the company’s particle-based multi-analyte technology. Levels of anti-cardiolipin and anti-beta 2 glycoprotein 1 IgG and IgM autoantibodies in human serum and citrated plasma are semi-quantitively determined.
Patients with antiphospholipid syndrome have an increased risk of blood clots. Also known as Hughes syndrome, the autoimmune disorder is characterised by the presence of antiphospholipid antibodies. Spain-headquartered Werfen says its reagents can be used to diagnose primary and secondary antiphospholipid syndrome with the help of other laboratory findings.
Werfen’s vice-president of research and development Michael Mahler said: “Early diagnosis is crucial in preventing complications as well as unnecessary procedures and increased healthcare costs. Aptiva APS IgG and APS IgM deliver expanded information to clinicians to help with the diagnosis and management of patients with autoimmune diseases.”
In October 2023, Werfen received US Food and Drug Administration clearance for a reagent to help diagnose connective tissue disease. Also in the company’s Aptiva range is a celiac disease test.
Aptiva can detect 19 CE-marked analytes, according to the company.
Werfen revealed that it has more than 60 further analytes in the pipeline, with more autoimmune diseases being targeted.
Werfen saw revenue of €2.2bn ($2.4bn) in 2022, down 0.7% from 2021. The company has had to deal with the decline of Covid-19-related sales but has grown its core businesses: Hemostasis, Acute Care and Autoimmunity.
In late 2022, the company completed its acquisition of US-based in vitro diagnostics company Immucor.
According to a market model by GlobalData, Werfen owns 10% of a diverse haemostasis in vitro diagnostic market. Roche and Abbott have the highest shares of 18.9% and 17.9%, respectively. The global haemostasis tests market is expected to grow to $675m by 2033.
Source: Werfen secures CE mark for antiphospholipid syndrome assays

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
