While the Omicron variant continues to infect people around the world, researchers at the University of Missouri have identified the highly prevalent, specific mutations that are causing the Omicron variant’s high rate of infection.

Ginkgo Bioworks said on Wednesday that it has acquired Project Beacon COVID-19, a Boston-based coronavirus testing organization.

ISB-led study shows unvaccinated pregnant people with COVID-19 are more likely to have poor birth outcomes even if they did not experience severe respiratory problems during infection.
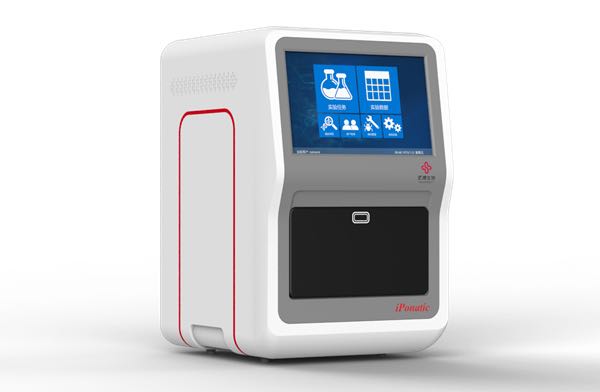
On January 4th, the four-channel iPonatic nucleic acid detection analyzer of Sansure Biotech was certified by FDA. This is another global authority certification obtained after the approval of China NMPA and the European Union CE certification.
The agency is expanding the use of a single booster of the Pfizer-BioNTech vaccine to children ages 12–15 and is shortening the time between primary vaccination and booster to at least five months.

Scientists at Scripps Research, University of Chicago and Icahn School of Medicine at Mount Sinai have identified a new Achilles’ heel of influenza virus, making progress in the quest for a universal flu vaccine.

Siemens Healthineers announces that the U.S. Food and Drug Administration (FDA) today granted Emergency Use Authorization (EUA) for the CLINITEST Rapid COVID-19 Antigen Self-Test,1, 2, 3 providing nationwide access to a new at-home or over-the-counter self-test as COVID-19 testing needs.
Scientists at Scripps Research, University of Chicago and Icahn School of Medicine at Mount Sinai have identified a new Achilles’ heel of influenza virus, making progress in the quest for a universal flu vaccine.

Scientists have identified unique “indicators” in the blood of patients with severe and fatal COVID, paving the way for simple diagnostic tests to help doctors identify who will go on to become critically ill.

Pfizer announced on Dec. 13, 2021 that it had entered into a definitive agreement to acquire Arena Pharmaceuticals, a clinical stage company specializing in therapies treating immuno-inflammatory diseases, for $6.7 billion dollars.
✔ All (518)
✔ Press release (3)
✔ Industry news (515)
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
