


At present, MGT-based blood group test card has been used extensively in the field of blood typing, and MGT boasts of easy operation, high sensitivity, accurate results that can be saved and other strengthens, which allowed it to become method recommended by American Association of Blood Banks (AABB), technique recognized by FDA and internationally recognized blood group analysis test technique.

Stay up-to-date with the latest happenings in the rapidly evolving field of In Vitro Diagnostics (IVD) in China.

Seegene, a global molecular diagnostics company, announced the European launch of its new antimicrobial resistance (AMR) testing product, Allplex™ MDRO Assay, which is CE marked under the EU In Vitro Diagnostic Regulation (IVDR). The assay is designed to support the detection of multidrug-resistant organisms (MDROs) associated with healthcare-associated infections (HAIs).

Foundation Medicine, Inc., a global, patient-focused precision medicine company, today announced that it has received approval from the U.S. Food and Drug Administration (FDA) for FoundationOne®CDx to be used as a companion diagnostic (CDx) for TEPMETKO® (tepotinib) developed by EMD Serono, the healthcare business of Merck KGaA, Darmstadt, Germany in the U.S. and Canada. TEPMETKO received accelerated approval from the FDA in February 2021 and traditional approval in February 2024 for the treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) harboring mesenchymal-epithelial transition (MET) exon 14 skipping alterations (METex14).

Diasorin (FTSE MIB: DIA) announced today that it has received 510(k) clearance from the US Food and Drug Administration (FDA) for the LIAISON PLEX Gastrointestinal Flex Assay, a fully customizable syndromic testing panel capable of detecting a market-leading 24 gastrointestinal pathogen targets. The new panel expands Diasorin’s growing LIAISON PLEX assay portfolio, which also includes a customizable respiratory assay and three blood culture panels.

Guardant Health, Inc. (Nasdaq: GH), a leading precision oncology company, today announced that the U.S. Food and Drug Administration (FDA) has approved Guardant360® Liquid CDx, advancing blood-based comprehensive genomic testing by integrating genomic and epigenomic insights and helping clinicians make better-informed treatment selection decisions for patients with advanced cancer. Guardant360 Liquid CDx is the largest FDA-approved liquid biopsy panel, assessing a 100X wider genomic footprint than the previously approved Guardant360 CDx to deliver comprehensive tumor profiling results. The seven previously U.S. FDA-approved companion diagnostic indications for Guardant360 CDx transfer to the new test with the FDA approval of Guardant360 Liquid CDx.

QIAGEN (NYSE: QGEN; Frankfurt Prime Standard: QIA) announced at the 2026 BIO-IT World Conference & Expo in Boston that the QIAGEN Digital Insights bioinformatics business and its curated knowledge bases and bioinformatics expertise will be integrating NVIDIA accelerated computing and the NVIDIA BioNeMo platform to help researchers use AI more effectively in drug discovery.
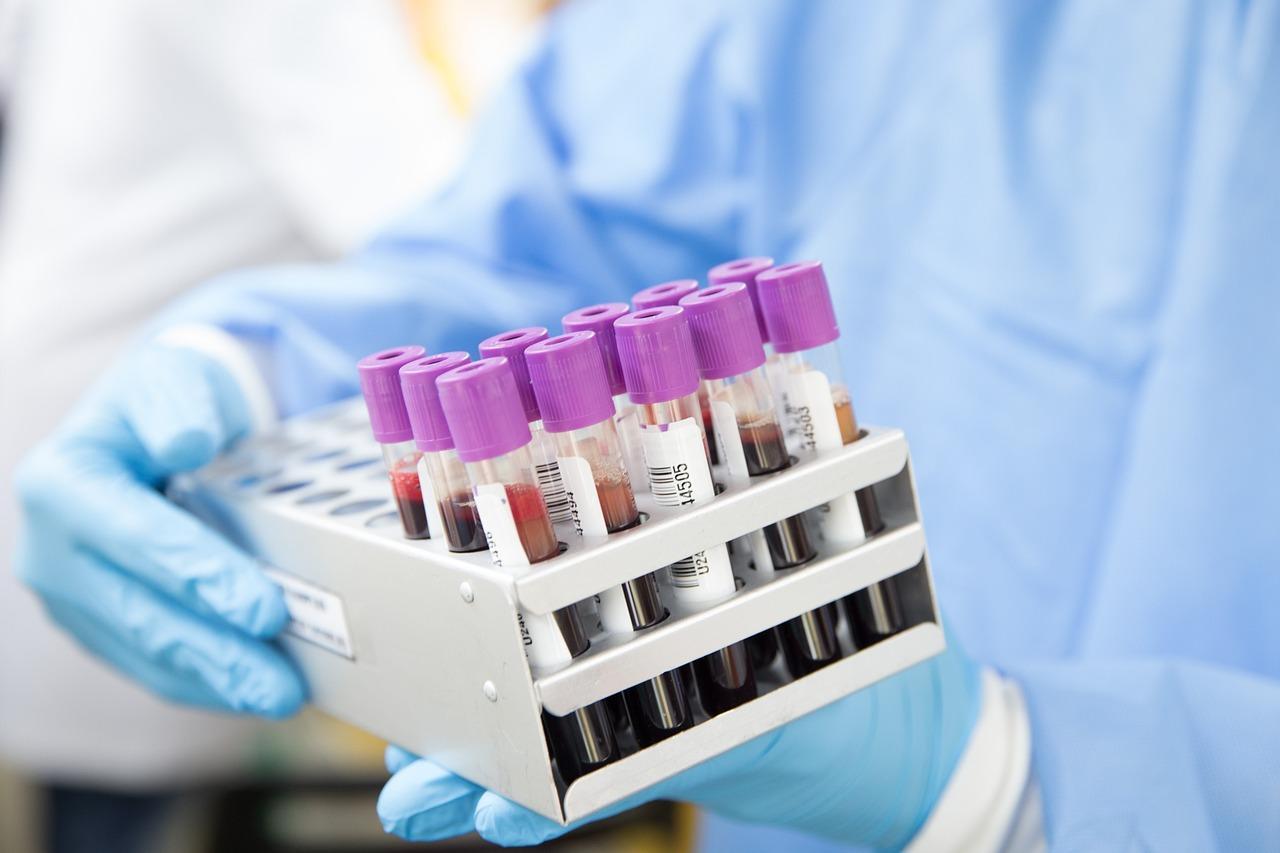
At present, domestic blood typing reagent-related products already covered ABO blood typing, test of Rh and other irregular antibodies, and anti-human globulin test.

AstraZeneca and Roche Diagnostics Asia Pacific today announced a three-year Memorandum of Understanding to help advance digital pathology capabilities and elevate cancer care across nine Asia markets, a first in the region. This landmark collaboration aims to accelerate the adoption of AI-powered digital and computational pathology through educational and training initiatives and improve biomarker testing in breast and lung cancer.

Stay up-to-date with the latest happenings in the rapidly evolving field of In Vitro Diagnostics (IVD) in China.

Co-Diagnostics, Inc. (Nasdaq: CODX) ("Co-Dx," or "the Company"), a molecular diagnostics company with a unique, patented platform for the development of molecular diagnostic tests, today announced its financial results for the first quarter ended March 31, 2026.

Natera, Inc. (NASDAQ: NTRA), a global leader in cell-free DNA and precision medicine, today announced that the U.S. Food and Drug Administration (FDA) has approved Signatera CDx as a companion diagnostic (CDx) for use with adjuvant atezolizumab (Tecentriq®) immunotherapy in MIBC.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
