


To ensure the quality and safety of medical services in primary healthcare institutions, China’s National Health Commission (NHC) and other relevant authorities issued a notice on 1 April 2026, outlining a three-year nationwide action.

Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Hologic, Inc. (Nasdaq: HOLX) announced today preliminary revenue results for its first fiscal quarter ended December 25, 2021.
To make fusion energy a viable resource for the world’s energy grid, researchers need to understand the turbulent motion of plasmas: a mix of ions and electrons swirling around in reactor vessels.
Ortho Clinical Diagnostics announced preliminary financial results for the fourth quarter after the close of the market on Thursday, saying it expects total revenues of between $520 million and $522 million, up approximately 1 percent year over year from $517 million.

Thermo Fisher Scientific, the world leader in serving science, and PeproTech, a leading developer and manufacturer of recombinant proteins, today announced that Thermo Fisher completed its acquisition of PeproTech on December 30, 2021 for a total cash purchase price of approximately $1.85 billion.

On January 7, Hotgen Biotech released a performance forecast. The company expects the net profit attributable to the shareholders of the listed company from January to December 2021 to be CNY 2 billion to CNY 2.35 billion, with a year-on-year change of 1684.65% to 1996.97%.

Illumina said on Wednesday that it has entered into a codevelopment agreement with SomaLogic to create multiplexed, high-throughput proteomics assays with next-generation sequencing-based readout.
When a patient receives a kidney transplant, doctors carefully monitor them for signs of rejection in several ways, including biopsy. However, this procedure is invasive and can only detect issues at a late stage.
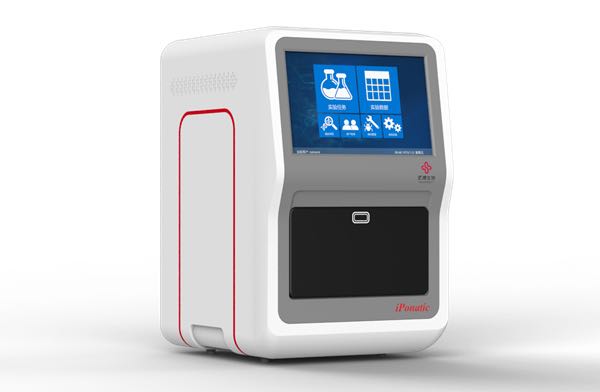
On January 4th, the four-channel iPonatic nucleic acid detection analyzer of Sansure Biotech was certified by FDA. This is another global authority certification obtained after the approval of China NMPA and the European Union CE certification.
Researchers in China have identified dozens of causal relationships between blood metabolites and the microbes in the human gut.

Calibre Scientific said on Tuesday that it has acquired French life science reagent and instrument distributor AmpliTech.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
